CDK4/6 Inhibitor
Abemaciclib
Shapiro GI1
Target
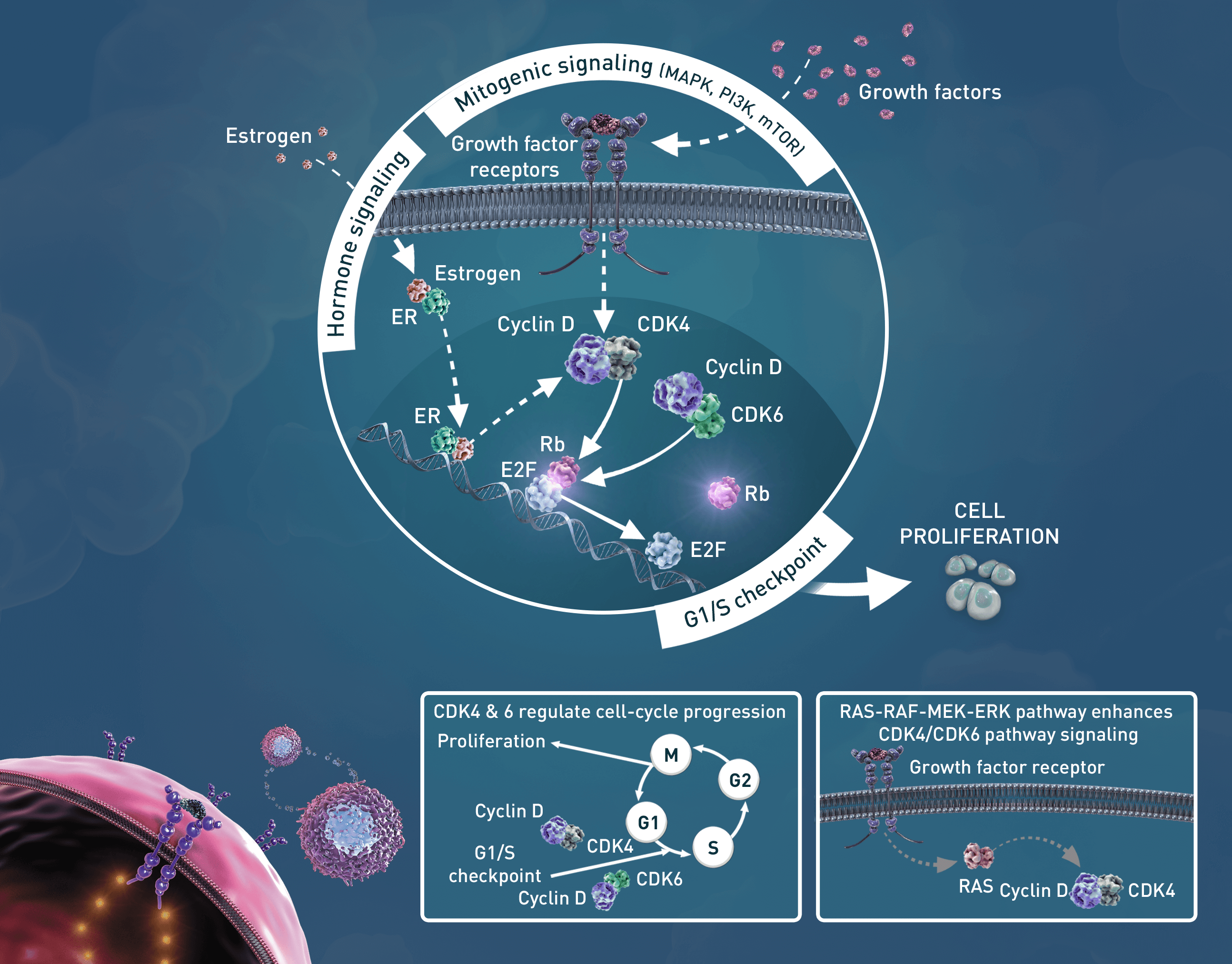
Many human tumors acquire alterations which can lead to the activation of cyclin-dependent kinases (CDKs). These alterations include mutations that directly activate CDK4/6; gene amplifications, which increase expression of various protein activators such as D-type cyclins; as well as genetic losses, which reduce expression of protein inhibitors such as p16. These various mechanisms as well as loss of retinoblastoma (Rb) can lead to an enhanced proliferative potential by decreasing dependency on external growth factors and mitogenic signaling pathways, which are required to stimulate growth under normal conditions.2,3
Molecule
Abemaciclib has been shown in vitro to be a selective ATP-competitive inhibitor of CDK4/6 kinase activity that prevents the phosphorylation and subsequent inactivation of the Rb tumor suppressor protein, thereby inducing G1 cell-cycle arrest and inhibition of cell proliferation.4,5
Clinical Development
Abemaciclib is being investigated in clinical trials in patients with breast cancer, non-small cell lung cancer, pediatric cancers, prostate cancer, or sarcoma.
References
- Shapiro GI. J Clin Oncol. 2006;24(11):1770-1783.
- Kim JK, Diehl JA. J Cell Physiol. 2009;220(2):292-296.
- Choi YJ, Anders L. Oncogene. 2014;33(15):1890-1903.
- Dempsey JA, et al. AACR Annual Meeting; April 6-10, 2013; Washington, DC. Abstract LB122.
- Gelbert LM, et al. Invest New Drugs. 2014;32(5):825-837.
or visit www.clinicaltrials.gov for more information on this trial
